TEPEZZA: a disease-modifying treatment that targets IGF-1R1-4
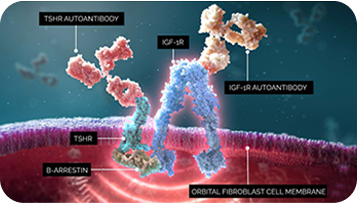

IGF-1R drives TED pathophysiology throughout the disease course—binding of autoantibodies activate the IGF-1R/TSHR signaling complex, activating orbital fibroblasts1,5,6
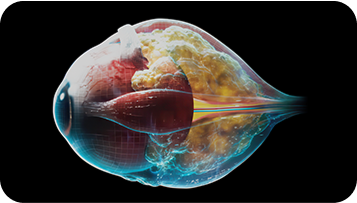

Orbital fibroblasts proliferate and cause expansion of fat and muscle tissue behind the eye2,3,5,7
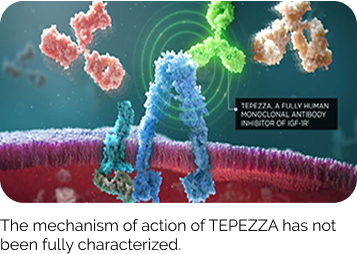

TEPEZZA targets IGF-1R, inhibiting orbital fibroblast activation at a root cause of TED1,5,6


TEPEZZA decreases proptosis by preventing fat and muscle remodeling and reducing tissue expansion behind the eye1,5,6
See TEPEZZA in action
Welcome, doctor.
You are here to learn how to identify and treat a rare and potentially debilitating disease affecting the human eye.
Although sometimes referred to as Graves’ orbitopathy, Thyroid Eye Disease, or T-E-D, is separate and distinct from Graves’ disease and has a unique underlying mechanism.
In Graves’ disease, autoantibodies target the thyroid-stimulating hormone receptor, triggering hyperthyroidism.
T-E-D is a chronic autoimmune disease with a heterogeneous presentation.
The reason for this difference lies deep within the human body.
Prepare to take a closer look.
In T-E-D, IGF-1R, a key mediator in the disease, and TSHR are colocalized and physically linked to form a functional signaling complex.
Central to the pathogenesis, autoantibodies activate the IGF-1R/TSHR signaling complex, which stimulates orbital fibroblasts, which can remain metabolically active throughout the course of the disease.
Once activated, these fibroblasts can cause potentially severe inflammation and expansion of muscle and fat behind the eye, leading to potentially debilitating downstream effects that are typical of T-E-D, such as proptosis and diplopia.
You have a treatment option available with TEPEZZA, the first FDA-approved treatment for T-E-D.
TEPEZZA (teprotumumab-trbw) is indicated for the treatment of Thyroid Eye Disease regardless of Thyroid Eye Disease activity or duration. TEPEZZA may cause infusion reactions. Infusion reactions may occur during an infusion or within one and a half hours after an infusion.
Please listen to additional Important Safety Information later in this video.
TEPEZZA targets and blocks IGF-1R and inhibit fibroblast activation via the IGF-1R/TSHR signaling complex.
IGF-1R inhibition on the surface of the orbital fibroblast is thought to attenuate downstream molecular events such as hyaluronan production, adipogenesis, muscle expansion, and inflammatory cytokine production.
Treatment with TEPEZZA has been proven to improve proptosis in T-E-D patients.
Congratulations, doctor, you now have the information to accurately identify T-E-D and understand the role of TEPEZZA in treating Thyroid Eye Disease.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
Infusion Reactions: TEPEZZA may cause infusion reactions. Infusion reactions have been reported in approximately 4% of patients treated with TEPEZZA. Reported infusion reactions have usually been mild or moderate in severity. Signs and symptoms may include transient increases in blood pressure, feeling hot, tachycardia, dyspnea, headache, and muscular pain. Infusion reactions may occur during an infusion or within 1.5 hours after an infusion. In patients who experience an infusion reaction, consideration should be given to premedicating with an antihistamine, antipyretic, or corticosteroid and/or administering all subsequent infusions at a slower infusion rate.
Inflammatory Bowel Disease: TEPEZZA may cause an exacerbation of inflammatory bowel disease (IBD). IBD has been reported in some patients without a prior diagnosis of IBD. Monitor patients for signs and symptoms of IBD. If IBD exacerbation is suspected, discontinue use of TEPEZZA.
Hyperglycemia: Increased blood glucose or hyperglycemia may occur in patients treated with TEPEZZA. In clinical trials, 10% of patients (two-thirds of whom had preexisting diabetes or impaired glucose tolerance) experienced hyperglycemia. Hyperglycemic events should be managed with medications for glycemic control, if necessary. Monitor patients for elevated blood glucose and symptoms of hyperglycemia while on treatment with TEPEZZA. Patients with preexisting diabetes should be under appropriate glycemic control before receiving TEPEZZA.
Adverse Reactions
The most common adverse reactions (incidence ≥5% and greater than placebo) are muscle spasm, nausea, alopecia, diarrhea, fatigue, hyperglycemia, hearing impairment, dysgeusia, headache, dry skin, ear discomfort, weight decreased, nail disorders, and menstrual disorders.
Please see Full Prescribing Information or visit TEPEZZAhcp.com for more information.
Talk to your Amgen representative today to learn more about TEPEZZA.
IGF-1R, insulin-like growth factor-1 receptor; TED, Thyroid Eye Disease; TSHR, thyroid-stimulating hormone receptor.


