The importance of completing full treatment course


8 IV Infusions
1 given every 3 weeks1
- Infusion 1: 10 mg/kg – 90 min1
- Infusion 2: 20 mg/kg – 90 min
- Infusions 3-8: 20 mg/kg – 60 min*
- TEPEZZA dosing is based on the patient’s actual weight
*If not well tolerated, the minimum infusion duration should remain at 90 minutes.
Find your patient’s dose
The below charts are intended to provide dosing calculations for each infusion stage for patients ranging from 40 kg to 182 kg. The dosing values can also be downloaded as a PDF.
Dosing Calculations
Determine the dosing and infusion values for your patient1
| Patient Weight | Infusion 1 (10 mg/kg) | Infusions 2 to 8 (20 mg/kg) | |||||
|---|---|---|---|---|---|---|---|
| lb* | kg* | Dose (mg) | Vials required (#) | Volume to withdraw (mL)† | Dose (mg) | Vials required (#) | Volume to withdraw (mL)† |
| 88 | 40 | 400 | 1 | 8.0 | 800 | 2 | 16.8 |
| 90 | 41 | 410 | 1 | 8.6 | 820 | 2 | 17.2 |
| 93 | 42 | 420 | 1 | 8.8 | 840 | 2 | 17.6 |
| 95 | 43 | 430 | 1 | 9.0 | 860 | 2 | 18.1 |
| 97 | 44 | 440 | 1 | 9.2 | 880 | 2 | 18.5 |
| 99 | 45 | 450 | 1 | 9.5 | 900 | 2 | 18.9 |
| 101 | 46 | 460 | 1 | 9.7 | 920 | 2 | 19.3 |
| 104 | 47 | 470 | 1 | 9.9 | 940 | 2 | 19.7 |
| 106 | 48 | 480 | 1 | 10.1 | 960 | 2 | 20.2 |
| 108 | 49 | 490 | 1 | 10.4 | 980 | 2 | 20.6 |
| 110 | 50 | 500 | 1 | 10.6 | 1000 | 2 | 21.0 |
| 112 | 51 | 510 | 2 | 10.7 | 1020 | 3 | 21.4 |
| 115 | 52 | 520 | 2 | 10.9 | 1040 | 3 | 21.8 |
| 117 | 53 | 530 | 2 | 11.1 | 1060 | 3 | 22.3 |
| 119 | 54 | 540 | 2 | 11.3 | 1080 | 3 | 22.7 |
| 121 | 55 | 550 | 2 | 11.6 | 1100 | 3 | 23.1 |
| 123 | 56 | 560 | 2 | 11.8 | 1120 | 3 | 23.5 |
| 126 | 57 | 570 | 2 | 12.0 | 1140 | 3 | 23.9 |
| 128 | 58 | 580 | 2 | 12.2 | 1160 | 3 | 24.4 |
| 130 | 59 | 590 | 2 | 12.4 | 1180 | 3 | 24.8 |
| 132 | 60 | 600 | 2 | 12.6 | 1200 | 3 | 25.2 |
| 135 | 61 | 610 | 2 | 12.9 | 1220 | 3 | 25.6 |
| 137 | 62 | 620 | 2 | 13.1 | 1240 | 3 | 26.1 |
| 139 | 63 | 630 | 2 | 13.2 | 1260 | 3 | 26.5 |
| 141 | 64 | 640 | 2 | 13.4 | 1280 | 3 | 26.9 |
| 143 | 65 | 650 | 2 | 13.7 | 1300 | 3 | 27.3 |
| 146 | 66 | 660 | 2 | 13.9 | 1320 | 3 | 27.7 |
| 148 | 67 | 670 | 2 | 14.1 | 1340 | 3 | 28.1 |
| 150 | 68 | 680 | 2 | 14.3 | 1360 | 3 | 28.6 |
| 152 | 69 | 690 | 2 | 14.5 | 1380 | 3 | 29.0 |
| 154 | 70 | 700 | 2 | 14.7 | 1400 | 3 | 29.4 |
| 157 | 71 | 710 | 2 | 14.9 | 1420 | 3 | 29.8 |
| 159 | 72 | 720 | 2 | 15.2 | 1440 | 3 | 30.2 |
| 161 | 73 | 730 | 2 | 15.3 | 1460 | 3 | 30.7 |
| 163 | 74 | 740 | 2 | 15.5 | 1480 | 3 | 31.1 |
| 165 | 75 | 750 | 2 | 15.7 | 1500 | 3 | 31.5 |
| 168 | 76 | 760 | 2 | 15.9 | 1520 | 3 | 31.9 |
| Patient Weight | Infusion 1 (10 mg/kg) | Infusions 2 to 8 (20 mg/kg) | |||||
|---|---|---|---|---|---|---|---|
| lb* | kg* | Dose (mg) | Vials required(#) | Volume to withdraw (mL)† | Dose (mg) | Vials required(#) | Volume to withdraw (mL)† |
| 170 | 77 | 770 | 2 | 16.2 | 1540 | 4 | 32.4 |
| 172 | 78 | 780 | 2 | 16.4 | 1560 | 4 | 32.8 |
| 174 | 79 | 790 | 2 | 16.6 | 1580 | 4 | 33.2 |
| 176 | 80 | 800 | 2 | 16.8 | 1600 | 4 | 33.6 |
| 179 | 81 | 810 | 2 | 17.0 | 1620 | 4 | 34.0 |
| 181 | 82 | 820 | 2 | 17.2 | 1640 | 4 | 34.5 |
| 183 | 83 | 830 | 2 | 17.4 | 1660 | 4 | 34.9 |
| 185 | 84 | 840 | 2 | 17.6 | 1680 | 4 | 35.3 |
| 187 | 85 | 850 | 2 | 17.9 | 1700 | 4 | 35.7 |
| 190 | 86 | 860 | 2 | 18.1 | 1720 | 4 | 36.1 |
| 192 | 87 | 870 | 2 | 18.3 | 1740 | 4 | 36.6 |
| 194 | 88 | 880 | 2 | 18.5 | 1760 | 4 | 37.0 |
| 196 | 89 | 890 | 2 | 18.7 | 1780 | 4 | 37.4 |
| 198 | 90 | 900 | 2 | 18.9 | 1800 | 4 | 37.8 |
| 201 | 91 | 910 | 2 | 19.1 | 1820 | 4 | 38.2 |
| 203 | 92 | 920 | 2 | 19.3 | 1840 | 4 | 38.7 |
| 205 | 93 | 930 | 2 | 19.5 | 1860 | 4 | 39.1 |
| 207 | 94 | 940 | 2 | 19.7 | 1880 | 4 | 39.5 |
| 209 | 95 | 950 | 2 | 20.0 | 1900 | 4 | 39.9 |
| 212 | 96 | 960 | 2 | 20.2 | 1920 | 4 | 40.3 |
| 214 | 97 | 970 | 2 | 20.4 | 1940 | 4 | 40.8 |
| 216 | 98 | 980 | 2 | 20.6 | 1960 | 4 | 41.2 |
| 218 | 99 | 990 | 2 | 20.8 | 1980 | 4 | 41.6 |
| 221 | 100 | 1000 | 2 | 21.0 | 2000 | 4 | 42.0 |
| 223 | 101 | 1010 | 3 | 21.2 | 2020 | 5 | 42.4 |
| 225 | 102 | 1020 | 3 | 21.4 | 2040 | 5 | 42.9 |
| 227 | 103 | 1030 | 3 | 21.6 | 2060 | 5 | 43.3 |
| 229 | 104 | 1040 | 3 | 21.8 | 2080 | 5 | 43.7 |
| 232 | 105 | 1050 | 3 | 22.1 | 2100 | 5 | 44.1 |
| 234 | 106 | 1060 | 3 | 22.3 | 2120 | 5 | 44.5 |
| 236 | 107 | 1070 | 3 | 22.5 | 2140 | 5 | 45.0 |
| 238 | 108 | 1080 | 3 | 22.7 | 2160 | 5 | 45.4 |
| 240 | 109 | 1090 | 3 | 22.9 | 2180 | 5 | 45.8 |
| 243 | 110 | 1100 | 3 | 23.1 | 2200 | 5 | 46.2 |
| 245 | 111 | 1110 | 3 | 23.3 | 2220 | 5 | 46.6 |
| 247 | 112 | 1120 | 3 | 23.5 | 2240 | 5 | 47.1 |
| Patient Weight | Infusion 1 (10 mg/kg) | Infusions 2 to 8 (20 mg/kg) | |||||
|---|---|---|---|---|---|---|---|
| lb* | kg* | Dose (mg) | Vials required (#) | Volume to withdraw (mL)† | Dose (mg) | Vials required (#) | Volume to withdraw (mL)† |
| 249 | 113 | 1130 | 3 | 23.7 | 2260 | 5 | 47.5 |
| 251 | 114 | 1140 | 3 | 23.9 | 2280 | 5 | 47.9 |
| 254 | 115 | 1150 | 3 | 24.2 | 2300 | 5 | 48.3 |
| 256 | 116 | 1160 | 3 | 24.4 | 2320 | 5 | 48.7 |
| 258 | 117 | 1170 | 3 | 24.6 | 2340 | 5 | 49.2 |
| 260 | 118 | 1180 | 3 | 24.8 | 2360 | 5 | 49.6 |
| 262 | 119 | 1190 | 3 | 25.0 | 2380 | 5 | 50.0 |
| 265 | 120 | 1200 | 3 | 25.2 | 2400 | 5 | 50.4 |
| 267 | 121 | 1210 | 3 | 25.4 | 2420 | 5 | 50.8 |
| 269 | 122 | 1220 | 3 | 25.6 | 2440 | 5 | 51.3 |
| 271 | 123 | 1230 | 3 | 25.8 | 2460 | 5 | 51.7 |
| 273 | 124 | 1240 | 3 | 26.1 | 2480 | 5 | 52.1 |
| 276 | 125 | 1250 | 3 | 26.3 | 2500 | 5 | 52.5 |
| 278 | 126 | 1260 | 3 | 26.5 | 2520 | 6 | 52.9 |
| 280 | 127 | 1270 | 3 | 26.7 | 2540 | 6 | 53.4 |
| 282 | 128 | 1280 | 3 | 26.9 | 2560 | 6 | 53.8 |
| 284 | 129 | 1290 | 3 | 27.1 | 2580 | 6 | 54.2 |
| 287 | 130 | 1300 | 3 | 27.3 | 2600 | 6 | 54.6 |
| 289 | 131 | 1310 | 3 | 27.5 | 2620 | 6 | 55.0 |
| 291 | 132 | 1320 | 3 | 27.7 | 2640 | 6 | 55.5 |
| 293 | 133 | 1330 | 3 | 27.9 | 2660 | 6 | 55.9 |
| 295 | 134 | 1340 | 3 | 28.2 | 2680 | 6 | 56.3 |
| 298 | 135 | 1350 | 3 | 28.4 | 2700 | 6 | 56.7 |
| 300 | 136 | 1360 | 3 | 28.6 | 2720 | 6 | 57.1 |
| 302 | 137 | 1370 | 3 | 28.8 | 2740 | 6 | 57.6 |
| 304 | 138 | 1380 | 3 | 29.0 | 2760 | 6 | 58.0 |
| 306 | 139 | 1390 | 3 | 29.2 | 2780 | 6 | 58.4 |
| 309 | 140 | 1400 | 3 | 29.4 | 2800 | 6 | 58.8 |
| 311 | 141 | 1410 | 3 | 29.6 | 2820 | 6 | 59.2 |
| 313 | 142 | 1420 | 3 | 29.8 | 2840 | 6 | 59.7 |
| 315 | 143 | 1430 | 3 | 30.0 | 2860 | 6 | 60.1 |
| 318 | 144 | 1440 | 3 | 30.3 | 2880 | 6 | 60.5 |
| 320 | 145 | 1450 | 3 | 30.5 | 2900 | 6 | 60.9 |
| 322 | 146 | 1460 | 3 | 30.7 | 2920 | 6 | 61.3 |
| 324 | 147 | 1470 | 3 | 30.9 | 2940 | 6 | 61.8 |
| 326 | 148 | 1480 | 3 | 31.1 | 2960 | 6 | 62.2 |
| Patient Weight | Infusion 1 (1.0 mg/kg) | Infusions 2 to 8 (20 mg/kg) | |||||
|---|---|---|---|---|---|---|---|
| lb* | kg* | Dose (mg) | Vials required(#) | Volume to withdraw (mL)† | Dose (mg) | Vials required(#) | Volume to withdraw (mL)† |
| 329 | 145 | 1490 | 3 | 31.1 | 2960 | 6 | 62.6 |
| 331 | 150 | 1500 | 3 | 31.5 | 3000 | 6 | 63.0 |
| 333 | 151 | 1510 | 4 | 31.7 | 3020 | 7 | 63.4 |
| 335 | 152 | 1520 | 4 | 31.9 | 3040 | 7 | 63.9 |
| 337 | 153 | 1530 | 4 | 32.1 | 3060 | 7 | 64.3 |
| 340 | 154 | 1540 | 4 | 32.4 | 3080 | 7 | 64.7 |
| 342 | 155 | 1550 | 4 | 32.6 | 3100 | 7 | 65.1 |
| 344 | 156 | 1560 | 4 | 32.8 | 3120 | 7 | 65.5 |
| 346 | 157 | 1570 | 4 | 33.0 | 3140 | 7 | 66.0 |
| 348 | 158 | 1580 | 4 | 33.2 | 3160 | 7 | 66.4 |
| 351 | 159 | 1590 | 4 | 33.4 | 3180 | 7 | 66.8 |
| 353 | 160 | 1600 | 4 | 33.6 | 3200 | 7 | 67.2 |
| 355 | 161 | 1610 | 4 | 33.8 | 3220 | 7 | 67.6 |
| 357 | 162 | 1620 | 4 | 34.0 | 3240 | 7 | 68.1 |
| 359 | 163 | 1630 | 4 | 34.2 | 3260 | 7 | 68.5 |
| 362 | 164 | 1640 | 4 | 34.5 | 3280 | 7 | 68.9 |
| 364 | 165 | 1650 | 4 | 34.7 | 3300 | 7 | 69.3 |
| 366 | 166 | 1660 | 4 | 34.9 | 3320 | 7 | 69.7 |
| 368 | 167 | 1670 | 4 | 35.1 | 3340 | 7 | 70.2 |
| 370 | 168 | 1680 | 4 | 35.3 | 3360 | 7 | 70.6 |
| 373 | 169 | 1690 | 4 | 35.5 | 3380 | 7 | 71.0 |
| 375 | 170 | 1700 | 4 | 35.7 | 3400 | 7 | 71.4 |
| 377 | 171 | 1710 | 4 | 35.9 | 3420 | 7 | 71.8 |
| 379 | 172 | 1720 | 4 | 36.1 | 3440 | 7 | 72.3 |
| 381 | 173 | 1730 | 4 | 36.3 | 3460 | 7 | 72.7 |
| 384 | 174 | 1740 | 4 | 36.6 | 3480 | 7 | 73.1 |
| 386 | 175 | 1750 | 4 | 36.8 | 3500 | 7 | 73.5 |
| 388 | 176 | 1760 | 4 | 37.0 | 3520 | 8 | 73.9 |
| 390 | 177 | 1770 | 4 | 37.2 | 3540 | 8 | 74.4 |
| 392 | 178 | 1780 | 4 | 37.4 | 3560 | 8 | 74.8 |
| 395 | 179 | 1790 | 4 | 37.6 | 3580 | 8 | 75.2 |
| 397 | 180 | 1800 | 4 | 37.8 | 3600 | 8 | 75.6 |
| 399 | 181 | 1810 | 4 | 38.0 | 3620 | 8 | 76.1 |
| 401 | 182 | 1820 | 4 | 38.2 | 3640 | 8 | 76.5 |
*Rounded to the nearest whole number.
†Rounded to the nearest tenth.
Saline bag size:
If dose is <1800 mg, use a 100-mL bag of normal saline (0.9% NaCl)1
If dose is ≥1800 mg, use a 250-mL bag of normal saline (0.9% NaCl)1
IV, intravenous.
In a post hoc analysis, 63% (n=15/24) of TEPEZZA patients who showed no proptosis improvement midway (12 weeks) through treatment ultimately did have a proptosis response after completing 8 infusions at 24 weeks from baseline.2
All TEPEZZA patients completing treatment had proptosis reduction3,*
Phase 3 study exploratory analysis: Change from baseline in proptosis over 24 weeks in individual patients
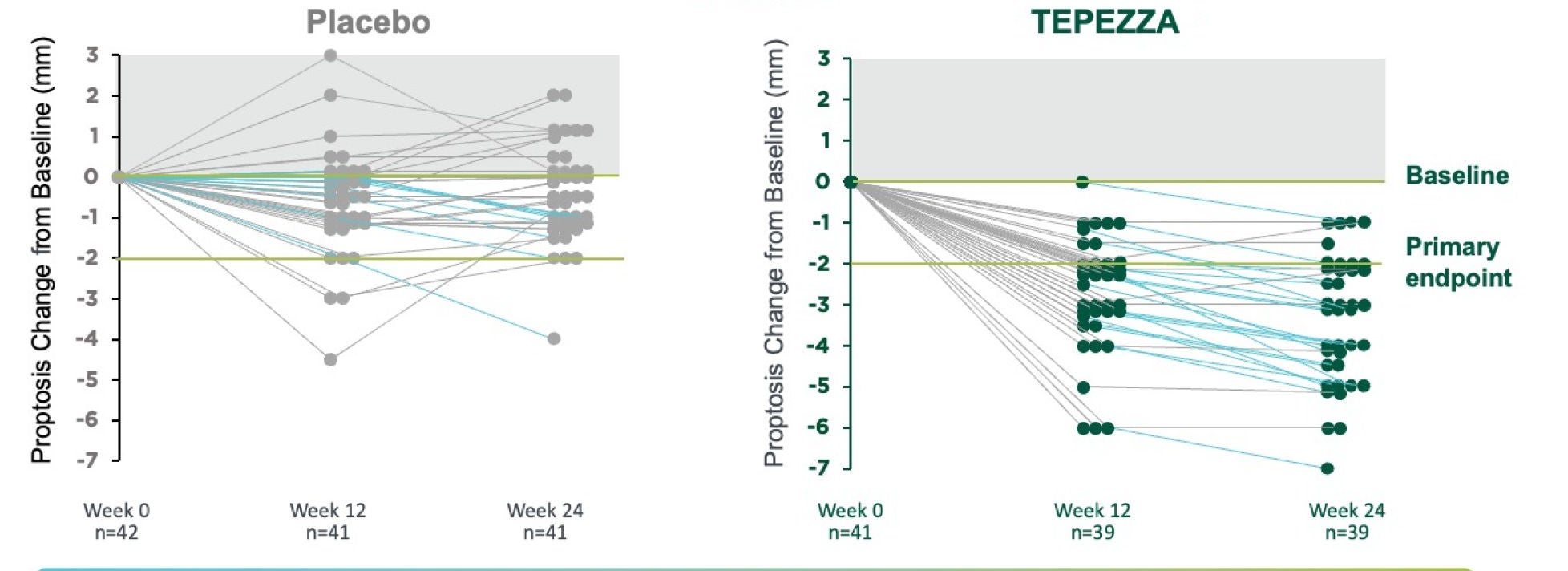


In a post hoc analysis, 63% (n=15/24) of TEPEZZA patients who showed no proptosis improvement midway (12 weeks) through treatment ultimately did have a proptosis response after completing 8 infusions at 24 weeks from baseline.2
*Analysis is exploratory and has not been adjusted for multiple comparisons. No conclusions of statistical or clinical significance can be drawn.

"After the second infusion, my symptoms seemed to decrease, and that motivated me to continue."
—Randi C., real TEPEZZA patient
In the Phase 3 study, improvement of proptosis as measured by mean change from baseline was observed as early as 6 weeks and continued to improve through Week 24.1
Nearly


OUT
OF


patients with TED would prefer IV medicine before trying eye surgery4
Based on a survey of 102 TED patients.



